Tue, Jul 22, 2025
[Archive]
Volume 21, Issue 1 (March - Special Issue 2024)
IJMSE 2024, 21(1): 8-15 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Patil A, Mahaparale S. Ferric and Ferrous Molar ratio Effect on Physical and Magnetic Properties of Iron Oxide Nanoparticles. IJMSE 2024; 21 (1) :8-15
URL: http://ijmse.iust.ac.ir/article-1-3532-en.html
URL: http://ijmse.iust.ac.ir/article-1-3532-en.html
Abstract: (12531 Views)
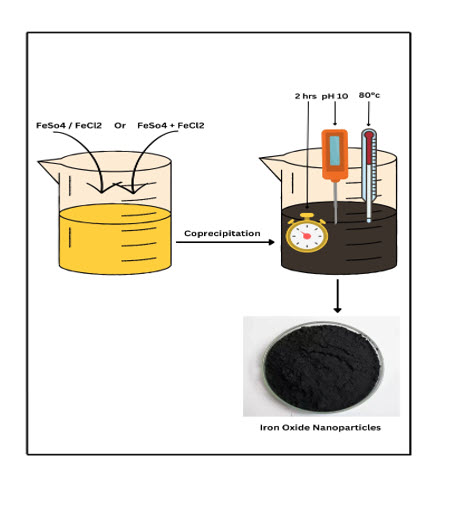
Iron oxide nanoparticles has attracted extensively due to their supermagnetic properties, preferred in biomedicine because of their biocompatibility and potential nontoxicity to human beings. Synthesis of iron nanoparticles (FeNPs) was prepared with the help of ferric chloride and ferrous sulphate by using the coprecipitation method. The variation and combination of ferric and ferrous concentrations affect the physical and magnetic properties of iron oxide nanoparticles. The effect of 0.1 M ferric and ferrous concentration on iron oxide nanoparticles studied separately and in combination. The obtained nanoparticles were characterized by Particle size, zeta potential, Ultraviolet (UV-visible), Fourier Transform Infrared Spectroscopy (FTIR), X-ray diffraction (XRD), Scanning electron microscope (SEM), Thermal gravimetric analysis (TGA), and Vibrating-sample magnetometer (VSM) techniques. Particle size was below 200nm and zeta potential was within the limit for all the batches. UV visible spectra at 224 nm, and FTIR exhibit two peaks at 510 and 594 cm-1, indicating iron oxide NPs and XRD confirmed the crystalline nature of Fe. SEM showed a spherical shape for all batches. The use of a combination of ferric and ferrous is more effective than its individual use. TGA and VSM studies confirmed its magnetic properties.
Type of Study: Research Paper |
Subject:
Biomaterials
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |